Mutations in the core RNA splicing factor SF3B1 are common across MDS, CLL, and clonal hematopoiesis. Prior studies have elucidated that mutations in SF3B1 result in neomorphic widespread changes in splicing due to usage of aberrant intronic branchpoint nucleotides. However, the molecular basis by which mutant SF3B1 induces mis-splicing is not established and therapeutic means to correct mis-splicing due to mutant SF3B1 do not exist.
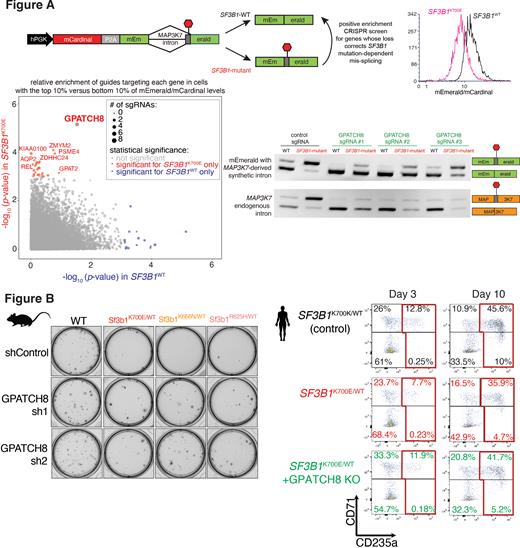
Recently, the aberrant gain-of-function splicing activity of mutant SF3B1 was harnessed to regulate expression of proteins in a mutant selective manner using synthetic version of endogenous mRNA sequences uniquely recognized by mutant SF3B1. Here, we utilized this technology to engineer a synthetic intron derived from MAP3K7 that interrupts the coding sequence of the fluorescent protein mEmerald such that mutant SF3B1 produces lower mEmerald expression than SF3B1 wild-type (WT) cells (Fig.A).
This fluorescent splicing reporter was then used to perform positive enrichment whole genome CRISPR screens to identify genes whose deletion corrects SF3B1 mutant aberrant splicing. GPATCH8 was the single most robust hit. GPATCH8 knockout strongly corrected mis-splicing of both synthetic and endogenous MAP3K7 introns (Fig.A). A previously unexplored protein, GPATCH8 has domains characteristic of RNA splicing factors including a G-patch motif, thought to be important in activating RNA helicases. Interestingly, recent work suggests that mutations in SF3B1 result in mis-splicing by disrupting physical interaction of SF3B1 to the RNA helicase DHX15 via the G-patch domain containing protein SUGP1. We therefore compared SUGP1 to GPATCH8 activity, evaluating first their transcriptome-wide binding sites using TRIBE-seq. We found that both proteins predominantly target intronic sequences over other genomic regions suggesting roles in splicing. RNA-seq of SF3B1 mutant and WT cells with or without GPATCH8/SUGP1 deletion revealed that ~33% of SF3B1 mutant splicing alterations are corrected by GPATCH8 deletion, while ~60% of SF3B1 mutant splicing alterations are recapitulated upon SUGP1 deletion. GPATCH8 regulated splicing events have stronger branchpoints indicating a potential role for GPATCH8 in 3' splice site recognition.
To further understand the connection between GPATCH8 and mutant SF3B1, we performed mass spectrometry studies of immunoprecipitated endogenous GPATCH8 in SF3B1 WT and mutant knockin cells. This revealed strong interaction between GPATCH8 and DHX15. GPATCH8 interacts with DHX15 at the same sites previously shown to be occupied by SUGP1 in crystal structures of the SUGP1/DHX15 interaction. These data and further biochemical studies elucidated that GPATCH8 competes with SUGP1 for interaction to DHX15. As such, deletion of GPATCH8 corrects SF3B1 mutant mis-splicing by enhancing the interaction of SUGP1 and DHX15 to the mutant SF3b complex.
Given that GPATCH8 is required for a large proportion of the splicing alterations induced by mutant SF3B1, we next evaluated the phenotypic effect of correcting SF3B1 mutant mis-splicing. We first tested the impact of anti-Gpatch8 shRNAs in bone marrow from WT mice as well as animals with conditional knockin of Sf3b1 K700E, K666N, or R625H mutations. While each Sf3b1 mutation results in impaired colony formation of hematopoietic precursors in methylcellulose assays, silencing of Gpatch8 rescued colony formation from hematopoietic precursors of each of the different Sf3b1 mutant mice and was tolerated by normal hematopoietic precursors (Fig.B). Moreover, while CRISPR base-edited knockin of the SF3B1 K700E mutation in adult CD34 + cells impaired erythroid development, this erythroid differentiation defect was rescued by GPATCH8 deletion in the same cells (Fig.B).
These data identify GPATCH8 as a novel RNA splicing factor involved in quality control of RNA branchpoint selection whose expression is required for mis-splicing by the different mutant forms of SF3B1. GPATCH8 antagonizes the activity of SUGP1 by competing for interaction with DHX15. These findings suggest that disrupting GPATCH8/DHX15 interaction could have important therapeutic benefit for the multitude of SF3B1 mutant hematopoietic diseases. This study also demonstrates the power of synthetic intronic splicing assays for discovery of trans factors and druggable proteins required by leukemia-associated mutant RNA splicing factors.
Disclosures
Abdel-Wahab:AbbVie, Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Loxo/Lilly: Consultancy; Nurix Therapeutics: Research Funding; Minovia Therapeutics: Research Funding; Amphista Therapeutics: Consultancy; AstraZeneca: Consultancy; Harmonic Discovery: Current holder of stock options in a privately-held company.